- Home
- Annual reports and publications
Annual reports and publications
The national research infrastructure Protein Production Sweden summarizes the year 2025 in the annual report. Annual reports from previous years are available in the navigation to the left. View the list of publications involving PPS via link below.
Annual report 2025
Protein Production Sweden (PPS) has in its fourth year consolidated its position as a national infrastructure.
Project applications are entered through the common web application form into our project database and are then discussed in the management group and distributed to the different labs. In this fourth year, 149 PIs applied for 235 different projects. This is a slightly smaller increase than from 2023 to 2024, perhaps indicating that we have reached most of the community interested in getting protein produced. Nevertheless, this shows a continued high demand for recombinant proteins from the Swedish scientific community.
During 2025, 205 projects were completed, resulting in the delivery of 472 protein batches (27 % increase compared to 2024). Our infrastructure has clearly gained momentum, but the high demand and increased delivery rate brings us to the edge of our capacities.
To continue spreading the information about PPS among Swedish researchers, we have presented our capabilities at different conferences and meetings on 24 different occasions.
In August 2025, PPS organized the yearly P4EU meeting for the European network of protein production facilities in Gothenburg, with 120 participants.
PPS received again very favorable user survey results. We have continued the implementation of the earlier decided consensus quality analyses of our proteins. Moreover, we have continued our work on several activities to develop our services.
PPS follows well the plan as given in the GanTT table.
We submitted an application for extension of PPS 2027-2030, and VR granted the application in September. The planned organization is similar to the present period, but plant cells will no longer be included. Moreover, we added a work package for joint development activities to improve PPS.
The management group has had regular bi-weekly digital meetings throughout the year. Several additional meetings took place in the fall, for budget discussions for the new period.
The steering committee has met five times, of which three meetings were with the renewed steering committee, who had their first meeting physically in Gothenburg in August 2025. The other meetings were on Zoom. The new committee is described in detail below.
The scientific advisory committee (SAC), with three members, had no meetings during 2025, but they were involved in discussions around the application for the new period late in 2024. SAC will be involved in the coming period when the new steering committee is also in full function.
In September, all PPS staff met for a fourth staff retreat with 27 participants at LINXS in Lund, who also hosted study visits to MAXIV and ESS.
It is still important to reach out with information about our existence and services, including new developments that we can provide. During 2025, we have reached out primarily by:
a) Our webpage with information about PPS and the online application system has been maintained with some major improvements. A LinkedIn page (currently 841 followers) for PPS has been maintained during 2025.
b) Members of the PPS management group have presented PPS at various occasions at national and international conferences, workshops as well as in educational context (for a full list, see appendix 1). The total number of outreach activities was 24 in 2025, slightly higher than last year.
c) Another important route for spreading the knowledge of PPS to the Swedish scientific community is through cooperation with other infrastructures. This continued in 2025. PPS is a stakeholder for the Integrated Structural Biology (ISB) platform of SciLifeLab, participated at the ISB User Day in Stockholm in December and at a two-day Integrative Structural Biology in Lund in October. PPS has also been presented at several SciLifeLab days at different geographical sites. PPS has produced proteins for several platforms within SciLIfeLab, including the DDD platform and Affinity Proteomics, as well as for the Fragmax platform and MAX IV.
d) A user survey with 34 questions was sent out to 260 applicants and PIs who had requested or received proteins from PPS in 2025. 42 replies (16%) representing users of almost all submodules, showed also this year that our users overall were very satisfied with PPS services, with a mean score of >4.6 (with 5 as the best) for services overall, the project communication, the project reporting, the project support and the protein product (Fig. 1). The lowest score, for “time from order to delivery”, reached 4.47 this year, a somewhat increased satisfaction also with this aspect as compared to previous years. The three largest research areas for the use of the delivered proteins were “Biomedical research”, “Molecular biology and biochemistry” and “Structural biology” (areas may be overlapping), and the three most common methods for which the proteins had been used were “Biochemical/biophysical assays”, “Immunological assays” and “FACS”.
Two questions from the PPS user survey for 2025 that got excellent grades:
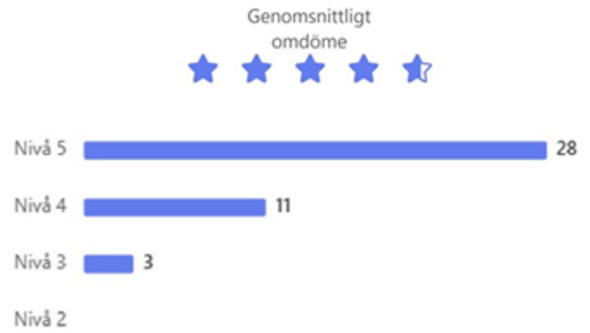
Figure 1: Overall, how satisfied were you with the services you got from PPS?
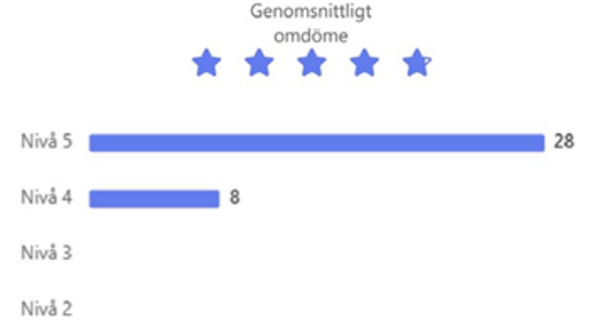
Figure 2: How satisfied were you with your protein product from PPS?
In module 3 (with its six submodules), the main part of the support and services in protein production are performed. This includes all steps from design of the expression plasmid, culturing of cells for the actual protein production in the expression system of choice, the purification and quality control. Module 4 contains our Gateway systems directed towards MAX IV and ESS.
Module 3.1 E. coli: The two E. coli-sites in Stockholm and Umeå saw a continued high national interest in their protein production services, with 160 applications and 261 protein batches delivered, corresponding to an increase of 10% compared to 2024. Both labs had staff replacements in 2024-2025 but are since March 2025 in full staffing again. Extensive work around the periplasmic production of proteins, through the testing of several signal peptides, was carried out at both sites. In Stockholm, two more projects with incorporation of non-canonical amino acids were performed, and in Umeå the focus has been the refolding and purification of proteins from inclusion bodies, and also studies on the effects of the bacterial strain, placement of the tag, temperature and culture medium on protein yield. Both sites have raised and continued to harmonize their user fees. The interaction between the two sites has resulted in knowledge-transfer, shared protocols and fruitful discussions on difficult projects.
Module 3.2 P. pastoris: This year there is a further increase of activity in the module, as compared to the previous year. The character of the YPP projects is that they are commonly very long, research-oriented, and connected to non-trivial production targets. During 2025, we had our first company project connected to a specific agreement and we evaluated the OPEN Pichia strain for production of a eukaryotic membrane protein. Yeast has a smaller number of proteins delivered but has the capacity to produce very high amounts (>500 mg) in successful projects. This year, YPP has delivered 15N-labelled BSA (65 mg) and there is an article in preparation where this protein product is used.
Module 3.3 Insect cells (BEVS): All three modules (module 3.1, 4.1 and 4.2) share the same laboratories and instruments as well as maintenance and support functions provided by LU. All three modules have successfully delivered intracellular and secreted proteins, protein complexes and membrane proteins to our users in numbers reaching the limit of our capacities (43 batches were delivered from module 3.3), and projects had to be selected according to PPS prioritization procedure. Nevertheless, we tested the use of fully defined chemical media for high density cell culture of insect cells according to this year’s operational plan.
Module 3.4 Mammalian cells: Protein production in mammalian cells has continued at both the Stockholm and Gothenburg sites, with a total of 78 protein batches delivered. This represents a 10% increase compared to 2024. Returning users from previous years have continued to engage with the platform, and new users are being added. Development activities outlined in the 2025 operational plan have progressed, with several initiatives already completed. In Gothenburg, expressed cytosolic proteins have been delivered in a user project and the isotope-labelling protocol is established, but still needs verification with more targets. Other activities related to protein purification are still being worked on at both sites.
Module 3.5 Plant cells: During 2025, Plant Cell Packs (PCPs) derived from A. thaliana cell suspension cultures were fully implemented, as a faster way to produce proteins (14 days). This allows for faster screening of protein constructs and optimal production conditions, and for transient protein production and the secretion of recombinant proteins. In addition to PCPs, co-cultures of A. thaliana cell suspensions and genetically engineered Agrobacterium containing the target gene(s) for infecting the plant cells are also pursued. Early in 2025, it was decided that the plant cell module will not be included in the application for extension of PPS, due to a low number of projects and low national interest. During 2025, two project requests came in and two protein batches were delivered.
Module 3.6 Cell-free Protein Synthesis (CFPS): In 2025, automated setup of CFPS reactions with the OT-2 liquid handler was optimized to also work in 384 well plates and for screening plasmid constructs of multiple proteins. The platform was used in several user projects and the number of batches delivered increased substantially from previous years. Automated one-step purification on the Agilent Bravo platform using Phytips was implemented as part of a user project, and this will in 2026 be extended to be able to do multiple step purification and/or protease cleavage with subsequent cleanup. 19F-labelling possibilities were explored as part of a user project, including incorporation of cytotoxic 19F-amino acids.
Module 4.1 (Per)deuteration and Module 4.2 Molecular chaperones: These modules together with module 3.3 constitute the Lund node of PPS. Besides many projects in both modules, development activities in 4.1 were done according to the operational plan for 2025 with establishing the use of a new bioreactor for deuteration and in 4.2 with testing of instrumentation using Flow Induced Dispersion Analysis for general QC of proteins and analysis of protein-protein complexes.
The PPS project database is growing and contained at the end of 2025 information about more than 900 proteins and protein constructs. The project application portal on the PPS website works without disturbances and is easy to use according to our user survey. Each PPS lab uploads their project reports for each completed project, together with DNA and protein sequences, expression system and whether production and purification were successful or not, to keep all data in one place in a uniform way.
For the PPS data to be fully searchable, we may however need to improve the format of this in the future, to make our data both FAIR and more useful, for example for the emerging area of AI-based predictive tools for protein expression and for protein design. These topics were discussed at the P4EU conference in August 2025, and initiatives are underway to find consensus on how such data should be collected. PPS follows this development closely.
The work continues according to the original plan and there are no major changes for this period. A new steering committee was appointed in the spring of 2025 (see below).
This year the steering committee for PPS has had five meetings, two with the old committee and three with the newly appointed committee.
In the beginning of the year, the application to VR for an extended period (2027-2030) was discussed and approved as was the annual report for 2024.
The first meeting with the new steering committee took place in Gothenburg in August. Marjolein Thunnissen (MAX IV, LU) continues as the chairman and the other members are Lovisa Holmberg Schiavone (AstraZeneca), Pär Nordlund (KI), Peter Nilsson (KTH, new), Christian Hedberg (UmU, new), Esmeralda Woestenenk (KTH, new) and Svend Kjaer (Francis Crick Institute, London, new). After the positive outcome of the application for extension, and following the budget discussions in the management group, the steering committee approved the budget for the new period. The budget and operational plan for 2026, the last year of PPS’ first funding period, were also approved in the fall.
The economy still follows the budget with no larger deviations, and 54% of the total costs so far have been covered by co-financing. The total costs for PPS decreased by 6% in 2025 compared to 2024. The total user fees showed an increase of 20% since 2024. The user fees were 67% higher than those budgeted for this year, even higher than last year.
The KPIs for PPS are defined as 1) the number of applied projects, 2) the number of completed projects, 3) the number of unique PIs for all applied and 4) completed projects and finally 5) the number of delivered protein batches (samples).
In 2025, PPS got 235 project applications, and 205 projects were completed. The projects came from 149 unique PIs and 135 PIs had completed projects. 472 protein batches were delivered, an increase of 26% since 2024.
There is a large demand for E. coli-production, making up 263 (56%) of the protein batches delivered. Production in mammalian cells gave 78 proteins delivered (17%) and BEVS 43 (9.1%). CFPS increased their deliveries this year to 70 (27% of all batches). They were mainly from a user project where automated purification was tried out.
An indicator for the national use of PPS is the proportion of projects that have been performed by another node than the users’ own universities. This number was found to be 56 and 54% for applied and finished projects respectively, continuing the small but positive trend towards a nationally distributed use of PPS (Figure 2). The applied and finished projects from universities that are not part of PPS were 21 and 20% respectively, a similar level to 2024.
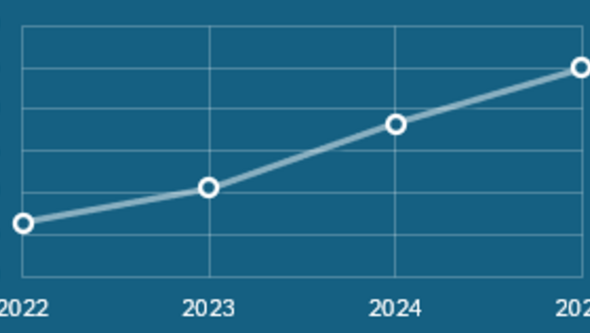
Figure 3: National use of PPS. Share of project applications (in %) that came from another university than the performing PPS node during 2022-2025.
The international recognition of PPS (28 applications) has increased compared to previous years (24 in 2023 and 21 in 2024 from foreign academics).
Projects from the industry sector increased to 14 in 2025 (compared to 4 in 2022, 6 in 2023 and 9 project applications in 2024) a small but continued positive trend.
The list of publications for 2025 contains 42 original research articles published by our users, compared to 36 last year, an increase with 17%. We now see acknowledgements to PPS in almost one third (31%, compared to 8% in 2023 and 28% in 2024) of the published papers. Many authors still acknowledge the individual platforms, probably connected to the time delay from production to publication. A full publication list is reported separately.
PPS had in December 2024 56% female employees, 43% women in both the management and the steering groups, and 67% women in the SAC. This quite even gender distribution continues at different levels within PPS and means no immediate need for action.
On the user side, we saw fewer projects from women (20%) and a reduced number of female PIs (20 and 23% for applied and finished projects, respectively). The corresponding values for previous years were all around 30%.
Only 13% of the protein batches were delivered to women (26% in 2024). We have no obvious explanation for this drop. The typical distribution of applicants based on project applications to VR in the areas medicine and technology is 25% - 50% women, and we see that as a reference value, which we do not fulfill this year. However, some projects with many delivered batches came from male PIs, for example the project using CFPS mentioned above, and this affected the low degree of batches to women.
A risk assessment was done as part of the original application, and it is considered by us as still valid. The identified risk that qualified personnel are leaving became evident in 2024, and this has affected productivity also this year. It is evident that the importance of offering an excellent working environment for the staff is still valid.
Along these lines, a fourth staff retreat was held in Lund in September 2025 and further yearly retreats are planned, the next in Gothenburg in September 2026.
We have also continued the group membership of the non-profit association Core Technologies for Life Sciences (CTLS) for all our staff.
PPS has earlier decided to follow consensus guidelines regarding quality control, and the agreed methods (SDS-PAGE to determine the purity, mass spectrometry to verify the identity, and analytical SEC/SEC-MALS/OMNISEC to determine homogeneity/monodispersity) are now routinely run for delivered purified proteins. This has proven not always to be possible due to the small size of the protein batches asked for/delivered, which makes it impossible to spare material for the quality tests (this is especially true for the analysis of monodispersity when SEC is not used as a second purification step). However, it remains a very important quality aspect for PPS to run the three core analyses that we have earlier agreed upon, and that this is included in the user fee. These efforts will be continued.
Workshops and conferences organized by PPS:
- Cell-free protein synthesis workshop, Gothenburg, 21-23 January (9 participants)
- 21st P4EU meeting, Gothenburg, 25-27 August (around 120 participants)
Oral presentations of PPS:
- Retreat for the Division of Protein Technology and Protein Engineering, KTH, February 2025
- GT-PRO PI meeting, March 2025
- D-MAP project meeting in Leiden, April 2025
- P4EU 21st Meeting, Gothenburg, August 2025 (PPS presentation)
- P4EU 21st Meeting, Gothenburg, August 2025 (selected talk from PPS staff)
- Master student course “Design and Production of Biomolecules” KEM350, Gothenburg, September 2025
- Workshop for Integrative Structural Biology, SciLifeLab Lund, October 2025
- Pro-PI Day, KTH, October 2025
- Presentation at Berner Lab workshop, November 2025
- Integrated Structural Biology User Day, SciLifeLab, Stockholm, December 2024
Posters or roll-up:
- MAX IV User Days, January 2025
- Research Day Umeå Centre of Molecular Research (UCMR), January 2025
- Sahlgrenska Academy Future Faculty symposium, February 2025
- Sahlgrenska Science Day, February 2025
- Wallenberg Centre for Molecular and Translational Medicine Science Day, April 2025
- SciLifeLab Lund node seminar, Lund, April 2025
- Lund spring symposium, May 2025
- LINXS-Loop opening, Lund, June 2025
- 27th Swedish Conference on Macromolecular Structure and Function, Tällberg, June 2025
- ICNS-Copenhagen, August 2025
- Chemical Biology Centre (KBC) Days, Umeå, November 2025
Other:
- Study visit of school class from Polhemsskolan’s Biotechnology Program, Lund, May 2025